Intellectual Property Rights Have Become A Key Part of Trade Deals
The protection and enforcement of intellectual property rights (IPR) have become an important component of current trade policy. For instance, the U.S.-China trade war that began in 2018 was influenced by American accusations of misappropriation of technology by China. And negotiations of the U.S.-Mexico-Canada Agreement, which replaced NAFTA in 2020, introduced a high-standard intellectual property (IP) chapter requiring strong and effective protection and enforcement of IPR.
The emphasis on IPR related to trade is not a new phenomenon. Indeed, the scope of regional trade agreements (RTAs) has changed over the recent decades. Prior to the formation of the World Trade Organization (WTO) in 1995, RTAs were mostly concerned with removing trade barriers between the countries signing the agreement. However, the Uruguay round introduced Trade-Related Aspects of Intellectual Property Rights (TRIPS) as part of overall trade deal negotiations. Since then, RTAs have begun containing substantial IPR provisions as part of their negotiations in most cases.
Evolution of RTA with IP Provisions
The exact details of the IPR provisions contained in each RTA can range from general provisions—covering things such as TRIPS reaffirmation, enforcement procedures, border measures and IPR exhaustion—to more specific provisions—referencing things such as copyright protections, trademarks, patents and even domain names. A particularly common subject included in IPR provisions of RTAs is provisions specifically concerning pharmaceuticals.
The figure below covers 437 enforced RTAs dating back to 1948. The agreements are split into three distinct categories:
- Those that do not mention IPR at all
- Those that mention IPR (e.g., only mentioned as a goal in the preamble) but do not contain any provisions relating to IPR
- Those that contain substantial IPR provisions
Classifications are taken from the National Swiss Science Foundation’s Design of Trade Agreements (DESTA) Database. The RTAs are split into six time periods: all RTAs prior to the formation of the WTO and then those that came into effect in subsequent five-year intervals until the last interval, which covers 2015-2020.
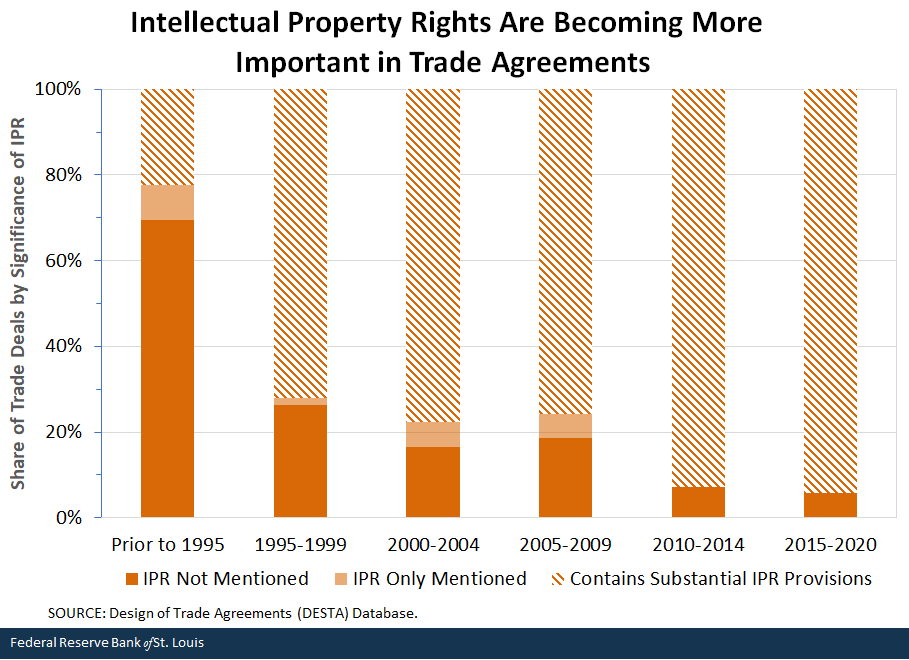
The trend is clear: 69% of RTAs enforced prior to the formation of the WTO did not mention IPR anywhere in the agreement. In contrast, since 2015, 33 of the 35 enforced RTAs contained strong IPR provisions; that is, 94% of agreements since 2015 contain such provisions. Notable among RTAs in this time frame was the failed Trans-Pacific Partnership (TPP). The TPP contained controversially restrictive IPR provisions that were pursued by the U. S. and that threatened to rewrite the global standards for IPR enforcement. When the U.S. withdrew from the agreement in early 2017, several of the stronger IPR provisions were dropped, and the agreement was signed by the remaining 11 countries as the Comprehensive and Progressive Agreement for Trans-Pacific Partnership (CPTPP).
The Importance of IPR Provisions for Developed Economies
Strong IPR provisions are typically required by developed countries to limit imitation and technology leakages in developing economies. IP-intensive industries have become increasingly important to the economics of developed countries. For example, in the U.S., IP-intensive industries account for 38.2% of the country’s gross domestic product; specifically relating to trade, IP accounts for 52% of all U.S. merchandise exports. As a result, it becomes increasingly important to developed countries to protect these industries in trade agreements with the rest of the world. Often this means incentivizing other countries to agree to provisions similar to what a country has domestically.
Conclusion
IPR as a part of trade agreements is at the forefront of economic debate once again. RTAs are increasingly containing strong IPR provisions, and trade wars have erupted partially over accusations of IP misappropriation and other related disputes. With the current COVID-19 pandemic, pharmaceutical IP has entered the discussion as well, as countries decide how to approach the distribution of medicines, vaccines and other medical-related IP across borders. As IP-related industries continue to become central to the economies of the world, there will need to be more research into the effects of these provisions and their impacts on the welfare of countries involved in agreements.
Additional Resources
Citation
Ana Maria Santacreu and Jesse LaBelle, ldquoIntellectual Property Rights Have Become A Key Part of Trade Deals ,rdquo St. Louis Fed On the Economy, June 14, 2021.
This blog offers commentary, analysis and data from our economists and experts. Views expressed are not necessarily those of the St. Louis Fed or Federal Reserve System.
Email Us
All other blog-related questions



